Glucose Restriction Fuels Metastatic Seeding: Tumor Glucose Deprivation Reprograms the Lung Innate Immune Landscape via Exosomal TRAIL
Overview and Key Highlights
On July 15, 2025, the team led by Professor Dong-Ming Kuang and Associate Professor Yuan Wei from the School of Life Sciences, Sun Yat-sen University, published an online research article entitled “Glucose Restriction Shapes Pre-Metastatic Innate Immune Landscapes in the Lung through Exosomal TRAIL” in Cell. By integrating large-scale clinical datasets, multi-omics analyses, and in vivo experiments, the study demonstrates that glucose deprivation can remodel the lung innate immune microenvironment via exosomal TRAIL to drive pre-metastatic niche formation, while also identifying potential intervention strategies that target this pathway.
Part One: Background, Motivation, Research Process, and Preliminary Findings
Background
Glucose metabolism is a cornerstone of tumor biology, providing a central energy source that supports rapid tumor cell proliferation. In recent years, strategies aimed at suppressing glucose metabolism, including low-carbohydrate diets, have received increasing attention as potential anti-cancer approaches. However, in cancer treatment, restraining primary tumor growth alone is often insufficient to improve patient outcomes; distant metastasis remains the leading cause of cancer-related mortality.
Glucose deprivation represents a major form of metabolic stress. A key unresolved question is whether restricting glucose metabolism may inadvertently shift tumor behavior toward a more malignant state. Emerging evidence suggests that targeting glucose metabolism not only constrains energy supply, but may also activate stress responses, reshape the tumor microenvironment, and influence immune regulatory programs. To date, systematic investigation remains limited regarding how glucose metabolic interventions affect distant immune landscapes and the formation of pre-metastatic niches.
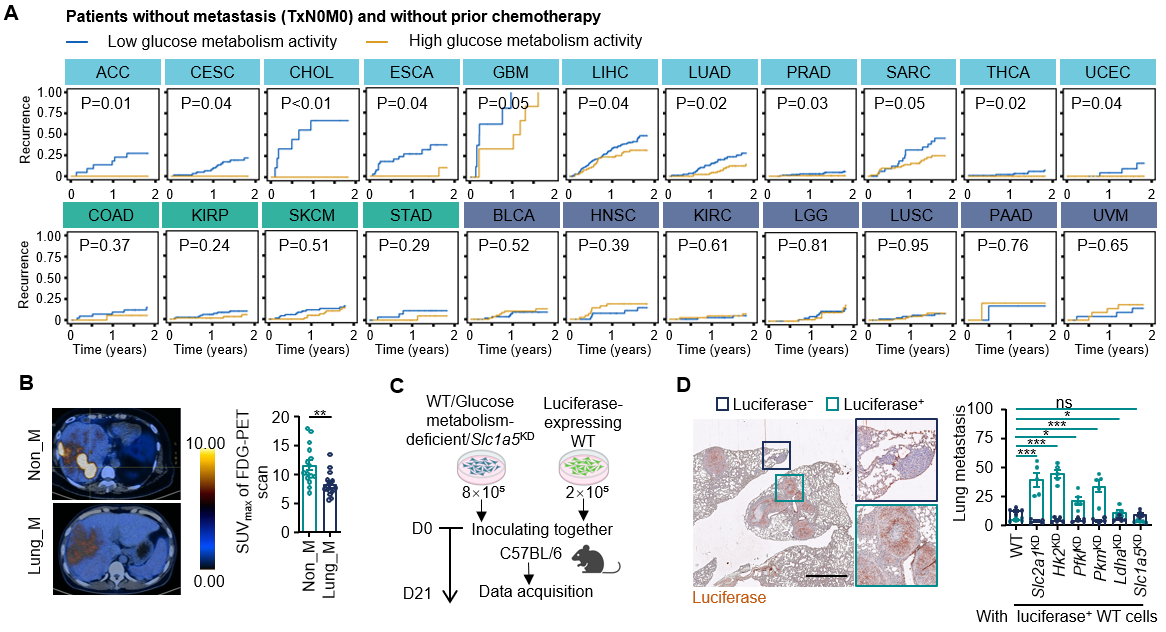
Impaired Glucose Metabolism Is Associated with Increased Metastatic Risk
The study analyzed large-scale clinical datasets spanning 15 cancer types and found that patients with lower tumor glucose metabolic activity exhibited a significantly higher risk of recurrence within two years after surgery. In hepatocellular carcinoma, tumors from patients who developed lung metastases exhibited significantly lower glucose metabolism than those without metastasis.
Consistent with these clinical observations, multiple mouse tumor models further showed that reducing tumor glucose metabolism, either through low-carbohydrate diets or genetic interventions, significantly increased lung metastatic propensity. Importantly, this increase in metastasis was not attributable to enhanced intrinsic migratory capacity of glucose-deprived tumor cells. Rather, the findings support a bystander effect, in which metabolically impaired tumor cells facilitate the metastatic spread of neighboring metabolically intact tumor cells.
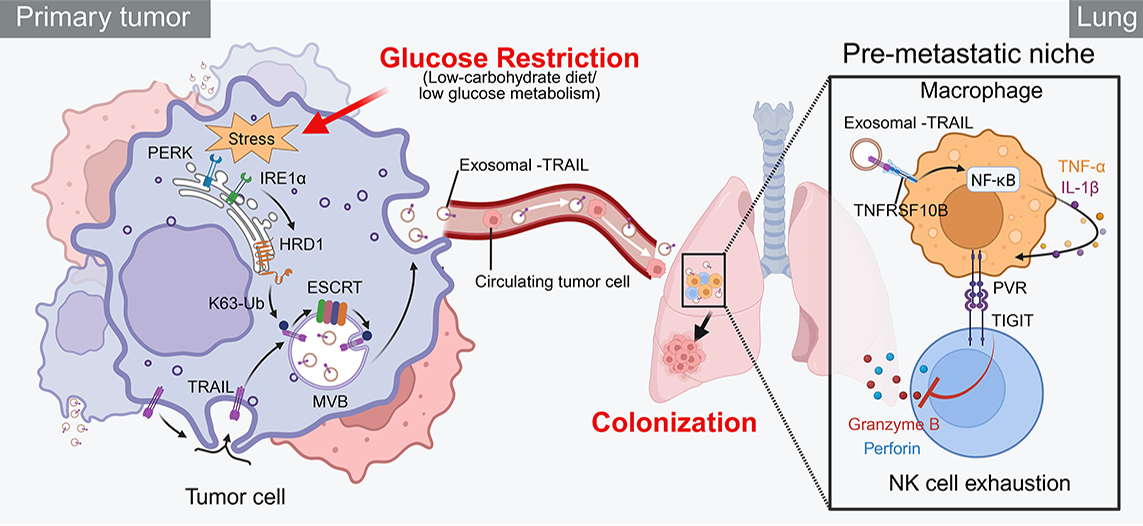
Mechanistic Insights: TRAIL-Mediated Immune Exhaustion Drives Pre-Metastatic Niche Formation
Mechanistic studies revealed that glucose deprivation induces endoplasmic reticulum stress in tumor cells, thereby activating the E3 ubiquitin ligase HRD1. This process promotes K63-linked ubiquitination of TRAIL and its subsequent sorting and packaging into exosomes via the ESCRT complex.
After entering the circulation and reaching the lung, TRAIL-containing exosomes drive the polarization of PVR⁺ macrophages, which suppress natural killer (NK) cell activity through the PVR–TIGIT axis, leading to NK cell exhaustion and the establishment of an immune microenvironment that supports tumor colonization.
Across multiple animal models, TIGIT blockade markedly reduced lung metastatic burden and also inhibited primary tumor growth, highlighting TIGIT as a potential therapeutic target in metabolism stress–associated metastasis.
Part Two:Research Challenges
This study spanned approximately seven years. Early in the project, the team observed an unexpected phenomenon: tumors with lower glucose metabolic activity were closely associated with postoperative recurrence—an observation that was counterintuitive and challenged prevailing assumptions. To validate this finding, the researchers systematically integrated clinical samples and pathological information, applying multiple approaches including RNA sequencing, lactate dehydrogenase (LDH) activity assays, spatial transcriptomics, and PET-CT imaging. These analyses consistently indicated that, in hepatocellular carcinoma patients who developed lung metastases, primary tumors exhibited significantly lower glucose metabolism than those in patients without metastasis. Complementary mouse experiments further showed that low-carbohydrate diets, while suppressing primary tumor growth, paradoxically enhanced distant lung metastasis.
This posed a conceptual challenge: conventional views emphasize that active glucose metabolism fuels tumor growth and dissemination, yet these results suggested that metastatic progression can increase under glucose restriction. This finding not only presents a theoretical paradox, but also challenges the prevailing paradigm that targeting glucose metabolism uniformly inhibits tumor progression. Despite skepticism, the research team remained convinced that a key mechanism underlay this unexpected observation.
Through extensive literature review, careful observation, and repeated analysis, the researchers recognized metabolicheterogeneity as a common feature of tumors, with regions of high and low glucose metabolism coexisting. This prompted a shift from viewing tumors as metabolically homogeneous entities toward considering functional interactions among distinct tumor cell populations. On this basis, the team hypothesized that glucose-restricted tumor cells may not be intrinsically more metastatic, but could indirectly promote lung metastasis by influencing neighboring metabolically normal tumor cells through a bystander effect.
To test this hypothesis, the team established and refined in vivo metastasis models and, through repeated validation, ultimately demonstrated that tumor cells with suppressed glucose metabolism can remodel distant immune environments through an exosome–immune axis, thereby promoting lung metastasis of metabolically intact tumor cells. This work deepens mechanistic understanding of the interplay among metabolism, immunity, and metastasis, and provides theoretical and experimental support for optimizing metabolism-targeted cancer therapies.
Part Three:Significance and Translational Potential
Starting from clinical observations and integrating multi-omics analyses with in vivo experimental evidence, this study systematically elucidates a mechanism by which glucose restriction promotes metastasis through an exosome–immune axis, and identifies the TIGIT pathway as a potential combinatorial therapeutic target. These findings have important implications for both basic research and clinical translation.
The findings underscore the need for careful evaluation of the systemic consequences of metabolic interventions in cancer therapy or prevention. For cancer patients, excessive glucose restriction may increase metastatic risk; for individuals with undiagnosed but potentially existing lesions, extreme low-carbohydrate diets could also create conditions permissive for tumor dissemination.
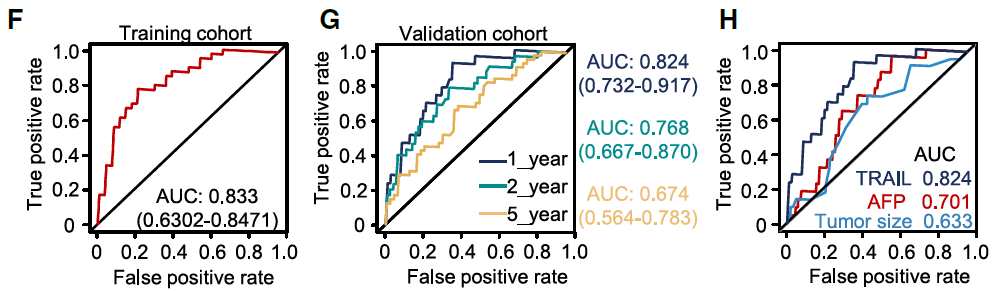
From a translational perspective, the team further showed that plasma exosomal TRAIL levels outperform conventional markers such as AFP and tumor volume in predicting postoperative lung metastasis in HCC patients, supporting its potential utility as an early risk stratification biomarker for clinical management.